Surgical Implants
Ellis Developments
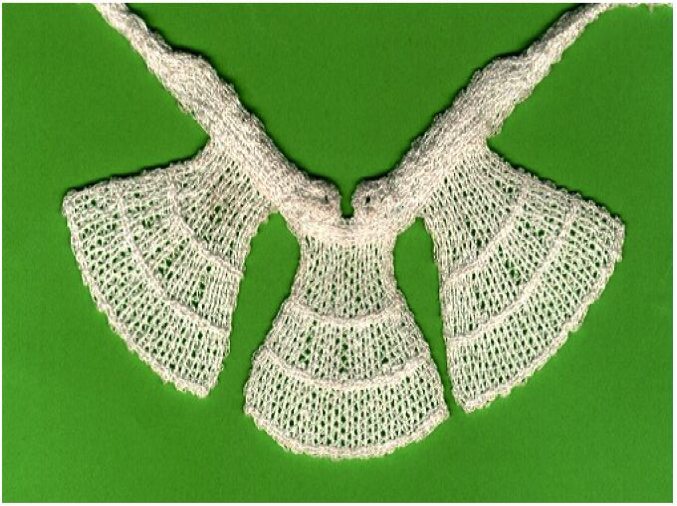
have developed a wide range of soft tissue implants. Some are still at a commercially sensitive stage, but others are described at each of the links above. There are two examples of what we can achieve in the shoulder surgery section .You will also find a picture gallery of a range of implants.
Spinal Surgery
Our embroidered implant systems for spinal disc replacement, Neodisc, have now been licensed, and are being further developed by NuVasive Inc., one of the world’s leading spinal companies.
Urology
Our main work in Urology has been within a £2.4m research and development project to look at improving the options for patients suffering from urinary incontinence. This major project, known as NuTaP, was part sponsored by the Department of Trade and Industry is the first major programme to look at the problem since the 1930’s. Our contribution to the programme was the development of a super-pubic port, to allow access to the bladder to ease drainage using an implantable tube running from the top of the bladder to a port in the region of the navel.
Aortic Root Surgery
We have devised a device to simplify the surgery to reinforce the area where the main aorta leaves the heart to supply the rest of the body. It is of particular importance to patients suffering from Marfan’s Syndrome.
Orthopaedics
We have a long experience (since 1981) in orthopaedic implants, and our advanced technology can assist in companies interested in further developments in knee and shoulder soft tissues in particular. We are, of course, interested in solving other orthopaedic problems too.
Shoulder Surgery
Ellis Developments has developed a number of devices in conjunction with the shoulder and Elbow Unit at Nottingham University and Nottingham City Hospital. The Nottingham Augmentation Device, designed for the reconstruction of large and massive Rotator Cuff tears has now been used on more than 100 patients with such tears. The results to date continue to be extremely good.
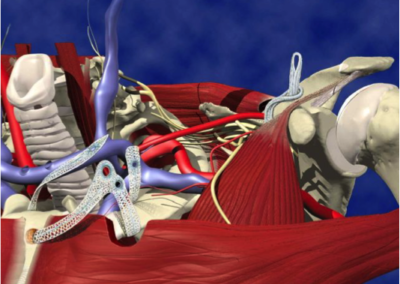
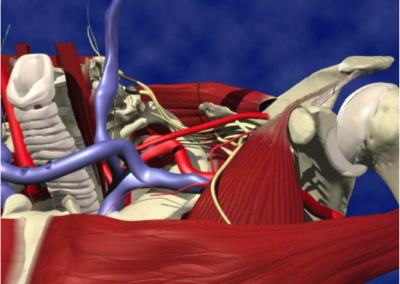
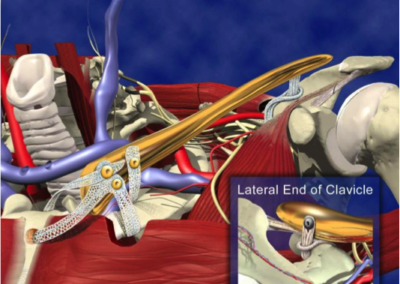
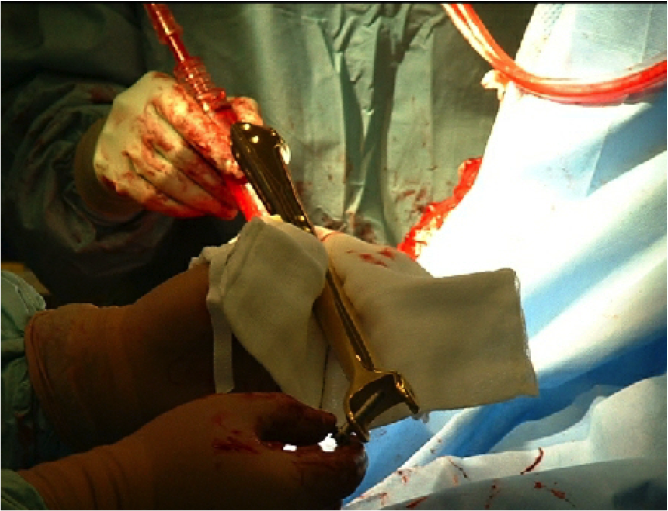
Claviculoplasty – Replacement of the Collar Bone
The world’s first claviculoplasty was performed in May 2003. The work to develop the implants used was collaboration between Ellis Developments, Pearsalls, Biomet and Surgicraft. The surgeon was Prof. W Angus Wallace at Nottingham City Hospital. This page tells of the contribution of all those involved.
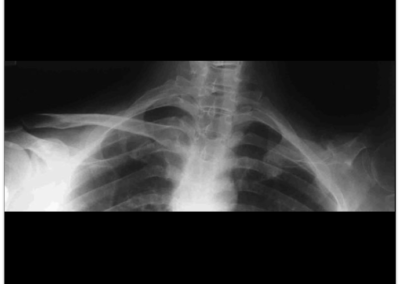
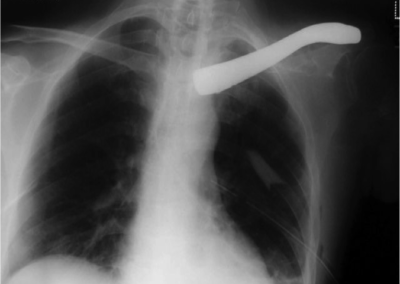
Before the operation the right hand side clavicle can be clearly seen on this x-ray, and below that the post-operative x-ray clearly shows the titanium implant.
In the development of endoprostheses, anatomical data plays an important role for the design and dimensioning of the implant. As Tim’s left clavicle had been previously excised it was planned to use the CT scan data from his right clavicle. This data was converted and transferred into a CAD system, where it was mirror imaged to produce the geometry of the proposed left clavicle.
This geometry was then utilised to produce a CAD model of the clavicle. At the Sterno-Clavicular end of the clavicle three spherical seated screws were positioned to allow the Sterno-Clavicular ligament to be attached. On the lateral posterior aspect of the clavicle, a small clamp plate and screw were positioned to allow the Acromio-Clavicular ligament to be attached.
With the design finalised the CAD model was then positioned into its anatomical position within the scan reconstruction, to check alignment and fit.
Titanium alloy was chosen for the implant, with a titanium nitride coating to improve wear resistance and help prevent tissue staining. All Biomet Merck implants are manufactured from proven materials and are fully content traceable and meet all relevant British, European and American standards.
Working closely with the engineers from Ellis Developments the design was finalised, and a wax model of the proposed clavicle prosthesis was sent to Professor Wallace for his approval.
CNC machining programs were written, and the clavicle was manufactured using highly skilled personnel and programmable computer- controlled machining centres, capable of producing implants to the finest tolerances.

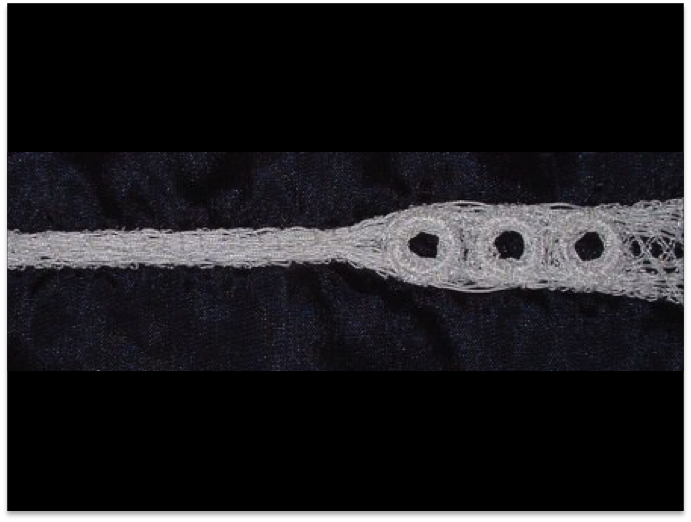
The Surgilig
Based in the West Midlands, Surgicraft was founded in 1924 and has a long and successful history of launching leading-edge medical devices. Surgicraft has developed a range of products across all of its main business areas, including the development and launch of the Surgilig in conjunction with Professor Angus Wallace and Mr Lars Neumann of the Nottingham Shoulder and Elbow Unit. The company also has a number of other products that are currently being launched in the UK and world-wide markets.
The Surgilig is a purpose-made artificial ligament designed to stabilise the collar bone following a severe dislocation. It secures the collar bone in place and is fixed with a stainless steel bone screw.
Surgilig utilises Surgicraft’s proprietary material technology to provide a strong biocompatible fixation that develops good tissue in-growth. It has been used nation-wide for the last three years in both NHS and private hospitals. The company has a considerable track record of developing innovative, leading-edge products for a number of diverse healthcare sectors and is currently finalising a private equity fundraising project to support the launch of its latest innovative shoulder product across Europe and other key export markets.
Embroidered Surgical Implants – Ellis Developments’ Involvement
This new technology was devised by Ellis Developments Limited and developed further through collaboration between Ellis Developments and Pearsalls Limited. Ellis Developments Limited is a Nottingham textile company, which specialises in the design and development of textiles for engineering applications, as well as surgical implants. Pearsalls is a very long-established Somerset company that manufactures suture thread as well as designing and manufacturing surgical implants made from suture threads.
The two companies have collaborated with Professor Wallace at Nottingham to produce repair systems for shoulders, and with Professor Brian Hopkinson at Nottingham to develop an artery liner to treat abdominal aortic aneurysms using keyhole surgery.
The technology to make the implant is a development from commercial embroidery techniques. The design is drawn up in Nottingham on a standard drawing package on the computer, the design transferred to a commercial embroidery design software package and the computer-based design programme prepared. The design is then e-mailed to Pearsalls in Somerset, where manufacturing can start, if necessary, within minutes.
The embroidery is carried out in a clean room using suture thread on a soluble cloth. Superimposed layers in the design ensure that the implant is strong and stable when the cloth is dissolved. The implant is sent for sterilisation by gamma irradiation and then despatched to the surgeon.
Prof. Angus Wallace – The Surgeon
Professor W Angus Wallace was appointed Professor of Orthopaedic and Accident Surgery at the University of Nottingham in 1985. He is Consultant Orthopaedic Surgeon to Nottingham City Hospital and the Queen’s Medical Centre and also Honorary Consultant to the Portland Training College for the Disabled in Nottinghamshire.
He has particular interests in shoulder and elbow surgery, biomechanics and passenger safety in transport accidents, sports medicine, medical informatics, osteoporosis and new methods of providing healthcare within the NHS.
His shoulder and elbow interests were highlighted when the Nottingham Shoulder and Elbow Unit was launched in August 1992. This is now one of the largest Shoulder and Elbow Units in the country and Prof. Wallace works alongside consultant colleagues Mr Lars Neumann and Mr Paul Triffitt.
Professor Wallace has been directly involved with the following innovations in the field of shoulder and elbow surgery:
- The Nottingham Total Shoulder Replacement system with an offset humeral head centre (Biomet Merck).
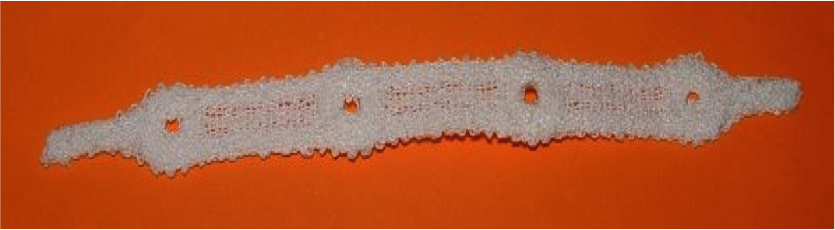
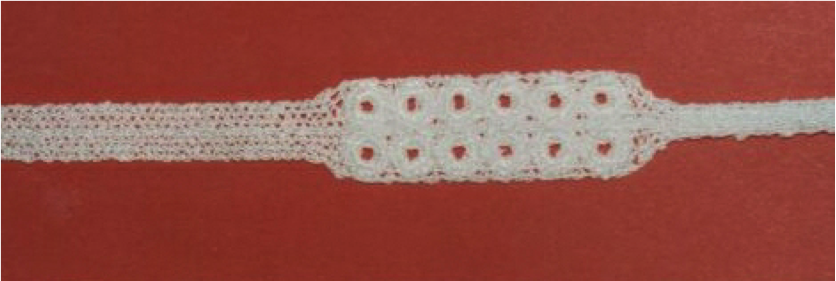
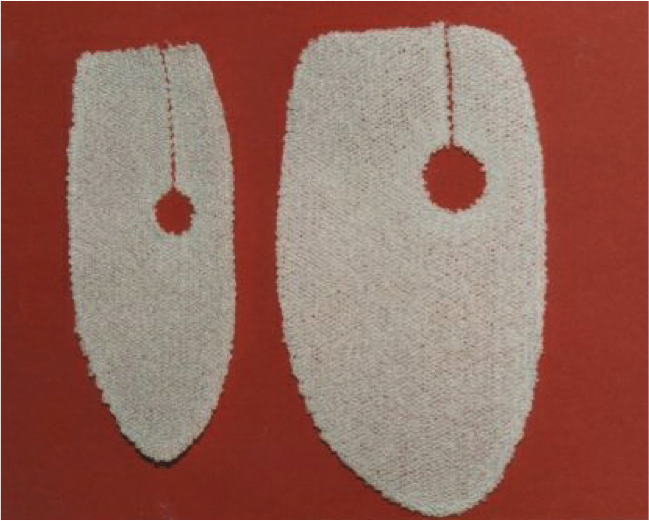
- The Nottingham Hood for use with shoulder replacements (Ellis Developments & Pearsalls).
- The Nottingham Surgilig for stabilising Acromio-clavicular joint dislocations (Surgicraft).
- The Nottingham Augmentation Device for the repair of tears of the Rotator Cuff Tendons and other indications (Ellis Developments & Pearsalls).
Professor Wallace holds many senior positions in shoulder and elbow surgery and biomechanics in the UK and worldwide, including President of the British Elbow and Shoulder Society and Co-Chair of the Research and Training Advisory Group, Medical Devices Faraday Partnership. He has received awards and prizes for his achievements in medicine and is a prolific writer and lecturer on orthopaedics.
Tim Baggaley – The Patient
Tim Baggaley is a freelance journalist, editor and designer with a career largely in magazine publishing and web design. Since his accident he has been developing a new career as an amputee actor after he was ‘discovered’ by a casting director at his weekly dance class. He secured a part in the current BBC 1 Saturday night horror series’ Strange’ as the demon ‘Kaa-Jinn’ and this has led to other opportunities, such as a new film ‘Shaun of the Dead’ when he plays a character which turns into a zombie and gets his arm chewed off. Tim is a keen motorsport fan, building motorcycles and preparing professional bike and car pit crews for races. He swims, fences and plays badminton and is an enthusiastic ballroom and Latin dancer – something he would like to go on to teach. He also enjoys theatre, play-reading and writing for pleasure. He writes poetry and gives recitals of his own work. More details about Tim and his career can be found on his website: www.timbaggaley.com
Back to top
Humerus Replacement
A Customised Implant Example
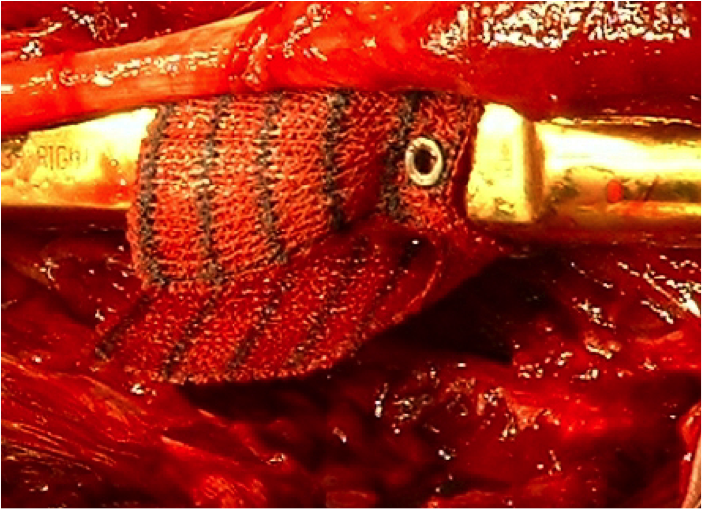
A patient with rheumatoid arthritis had a replacement shoulder and a replacement elbow, but the humerus fractured in the region between the two prostheses. A replacement customised implant was prepared by Biomet for Prof. Angus Wallace at the Shoulder and Elbow Unit at Nottingham City Hospital. The soft tissues needed reattaching to the metal component, therefore we designed a custom device, using as a starting point the rotator cuff repair device known as «The Nottingham Hood” This was screwed to the metallic implant to reattach the Deltoid muscles to the middle of the humerus, and a standard Nottingham Hood was used to reattach the rotator cuff at the shoulder.
Back to top
The Nottingham Hood (Presentation)

Back to top
Back to top
Vascular Surgery
In a major project, to a value of £320,000, half funded by a grant from the Department of Health, we developed a repair system for the keyhole surgical repair of abdominal aortic aneurysms.
Embroidery Technology is not exactly new. Hand embroidery has been used for decorating textiles for thousands of years; even automatic embroidery machinery has been in use for over a 150 years. The only thing that is really new is its application to surgical implants, which has been developed within the last few years.
Textile surgical implants have, until now, been constrained by the use of the traditional methods of knitting, weaving or braiding to make fibre assemblies. However, weaving usually limits the designer to placing fibres interlaced at right angles to one another, knitting forms the fabrics in loops which are generally dimensionally unstable, and braiding confines the designer to (usually) narrow structures made in a process similar to plaiting hair.
Embroidery is the formation of stitches on a base cloth, it being possible to place the stitches in any position. The fibres between stitches can thus be in any direction desired. The base cloth can be dissolved away after the stitching process has been carried out, and, assuming appropriate design features have been incorporated, the structure holds securely together as a stable entity. When making technical textiles, one is usually interested in structure rather than appearance and since fibres can be placed in any orientation in the plane of the base cloth, endless possibilities arise, especially mimicking natural fibrous arrays such as in natural ligaments. For example, the fibres in a particular ligament may fan out from a bundle 3 mm wide to an array 20 mm across, a design feature, which is easy to reproduce with textile fibres using embroidery technology. Another example could be a fracture-fixation device which has load-bearing threads arranged optimally to fit the part of the body where it is to be used and with open mesh parts which allows tissue ingrowth and consequent incorporation into the body.
The colour change facility of the machine can be used to alter the components in various parts of the implant as it is being manufactured – and allows the automated incorporation of attachment parts and other features very easily.
Modern embroidery uses sophisticated software dedicated to the quick and easy production of designs for the decoration of garments. Modifications to the use of this software allow fibre arrays to be designed for use as surgical implants with similar facility. This leads to the possibility of customised implants for individual patients. By way of example, we have taken a rough sketch of a component from a surgeon and converted it into an embroidery design, e-mailed it to Pearsalls Limited in Taunton, Somerset, and moved from concept to a preliminary concept manufactured product in less than three hours. The potential, even for specialist trauma surgery, is very exciting.
We have proved the general concept of using embroidery for surgical implants in a MedLINK project, which developed a device for the endovascular repair of abdominal aortic aneurysms. Although up to 1900 different size combinations are required to treat all the patients likely to be seen by a surgeon, special software now allows us to take the dimensions of an aneurysm provided by the surgeon from CT Scans and produce a customised embroidery design within seconds, and commence manufacturing within minutes. The device has significantly improved characteristics when compared with most other available devices.
Other work continues to be carried out to develop a range of orthopaedic implants for a diverse range of end users. All the devices in advanced stage of development at the moment are mainly constructed of polyester suture thread. One of the reasons for this is that the use of threads produced specifically for surgical applications raises no questions for surgeons about suitability for well designed implants: their behaviour and properties in the body are well documented. However, we are rapidly developing new advanced materials and structures and the development of active implants. The future is very encouraging and it is hoped that the technology will help in producing the framework for organs using the patient’s own cultured tissue which will replace diseased and damaged parts without the problems currently associated with transplant surgery.
The device developed, in collaboration between Ellis Developments, Pearsalls Ltd, of Taunton, Somerset, Prof. Brian Hopkinson of The University of Nottingham and Lombard Medical has resulted in a highly successful endovascular graft marketed by Lombard.
Abdominal Aortic Aneurysms and Embroidered Implants
Abdominal aortic aneurysms (AAA’s) are responsible for the death of some 10,000 people each year in the UK alone. These swellings in the wall of the main aorta are liable to rupture, with massive blood loss into the abdominal cavity.
The condition is more common in males, with approximately 5% of men over the age of 60 developing them. For the last 40 years the conventional treatment has been to open the abdomen to remove the diseased portion of the artery, and sew a tube of polyester in its place. However, the operation is risky for a number of patient groups, requires a long hospital stay, usually including several days in intensive care.
In recent years surgeons have been developing minimally invasive techniques for the repair of AAA’s. This usually requires a small opening in the leg to obtain access to the femoral artery, and uses an X-ray unit with image intensifier to monitor the progress of the procedure. Because the trauma to the patient is less intense, the level of hospital aftercare is considerably reduced.
The major research and development project we managed, was known as the SITE project (Surgical Implants using the Techniques of Embroidery). Details can be seen by following the link above.
Embroidered Implants
The UK Department of Health has for many years supported research and development in health devices. Their current programme, known as Invention for Innovation (i4i), supports collaborative research projects in a very wide range of devices ranging from surgical implants to walking aids.
The previous programmes known as MedLink and Health Technology Devices (HTD) had slightly different objectives but provided 50% funding support for projects. The scheme provided an ideal support process for a project that Ellis Developments Ltd set up and managed.
The Professor of Vascular Surgery at the University of Nottingham, Brian Hopkinson had been greatly interested in the repair of abdominal aortic aneurysms using minimally invasive (keyhole) surgery for a number of years. However, he was not entirely satisfied with the devices being developed, so he and I discussed the requirements of his ideal device one evening at his home. Ellis Developments had, already developed the concept of using embroidery technology to produce customised surgical implants and using the technology to produce graft stents was a process ideally suited to its facility for producing customised devices. Work carried out by Professor Hopkinson’s team had indicated that a device to fit all the patients they were likely to treat would need some 1900 different shapes and sizes of device to be produced. It was clearly impossible to produce this range using conventional weaving or knitting techniques, but embroidery provides the flexibility to produce customised devices.
Commercial embroidery processes have been developed to facilitate the production of very low numbers of customised devices. For example, embroidered name tags on company uniforms and other garment decoration. A commercial embroidery machine is a development of a simple lockstitch sewing machine with a computer controlled pantograph moving the work piece under the stitching head with great precision. Software packages for designing for the machines are readily available and their adaptation for use for surgical implants did not require any changes to the software.
Aortic aneurysms are weaknesses in the walls of a major blood vessel which, if they burst, cause internal bleeding within the patient. Aneurysms in the main artery, the abdominal aorta, give rise to some 10,000 deaths each year in the UK alone. Traditional treatment for several decades has been to use a woven (or sometimes knitted) polyester tube. In a major operation the diseased artery is clamped above and below the diseased portion, the diseased section removed and the polyester stitched into its place. . The patient population is normally older people, who often have other disease, and the operation has a high post-operative mortality rate. The benefit of using minimally invasive techniques is that they are less stressful to the patient and can sometimes even be done under a local anaesthetic, avoiding the need for a more dangerous general anaesthetic.
Keyhole surgery for the repair of abdominal aortic aneurysms involves the surgeon making a small incision in the femoral artery at the top of the leg and pushing a 7 mm catheter up through the blood vessels to reach the diseased section. The whole operation is carried out using x-ray techniques with image intensifiers to assist the surgeon in locating the position of the insertion system. Originally the repair devices known as a stent grafts, were pulled through the catheter. However, as the process has developed more sophisticated and easier to use insertion devices have been developed.
Although the insertion catheter is only 7 mm in diameter, the implant may be required to expand within the patient to a diameter of 34 mm or even larger: the main aorta carries a considerable amount of blood.
In order to provide the springiness required to incorporate this expansion we decided to use a superelastic wire made of nitinol (an alloy mainly of nickel and titanium which also has shape memory properties). The springiness is not only required to provide the self-expansion properties of the device, but also provide a blood-tight seal against the wall of the artery at both the top and the bottom of the device, which bridges the diseased section of artery.
A collaboration was set up between the University of Nottingham, Pearsalls Limited (who are the licensee of our embroidered implant process technology) and Lombard Medical of Didcot, Oxfordshire. We applied to the MedLink scheme for a project of a value of some £320,000, of which half was funded by the government. We found that one of the major benefits of receiving government funding was not just the financial support but also the support and contact given by the representative of the programme management committee and his extensive contacts.
Much of the initial development work was based on the design of the positioning of the shape memory alloy wire, which was attached to a 100% polyester micro fibre fabric. The configuration of the wire on the base fabric is critical in obtaining not only a fabric which will expand and provide a blood tight seal as required but will also prevent the tubular structure from kinking as the patient moves about and therefore obstructing the flow of blood from the heart towards the leg.
As work progressed studies were important in confirming the feasibility of inserting the device reliably and accurately inside the patient.
By the time the project ended (which took about 3 years), we had a device which we believed to be safe from a biocompatibility point of view as well as being safe for insertion into patients. A steady roll out programme is in hand where the pioneer surgeon, Professor Hopkinson teaches surgeons and later supervises them in the insertion of devices. The surgeons he trains can then teach other centres so the device is steadily rolled out across the world.
We expect Lombard to have considerable commercial success with this device in saving the lives of very many patients.
click here to go back to image
Embroidered Surgical implants
Surgical Implants using the Techniques of Embroidery
This report describes our breakthrough research into our patented method for the rapid design and manufacture of textile surgical implants using embroidery.
Project Report Prepared on behalf of the Project
Collaborators by:
Julian Ellis
Project Manager
Summary
- Introduction
- The Demonstration component
- Embroidery Techniques
3.1 Cornely
3.2 Schiffli
3.3 Lockstitch
- Embroidery Software
- Endovascular Aneurysm Graft Stent Specification
- Implant Materials
- Reinforcement Patterns
- Processing
- Project collaborators
9.1 Ellis Developments
9.2 Pearsalls Sutures
9.3 Division of Vascular Surgery, University of Nottingham
9.4 Anson Medical Limited
9.5 Contributions of each Partner
9.6 Interaction of collaborators
- Design Control
- Endoleak Research
- Cost Evaluation
- Achieved Project Deliverables
14 Dissemination
14.1 Journal Papers
14.2 Published Conference Papers
14.3 Conference Presentations
14.4 In-Preparation
14.5 Others
14.6 Patents
- Exploitation
15.1 Achievements and Further Plans
- Parallel Projects
16.1 A device for the Repair of the cervical spine.
16.2 Spinal Ligament Prosthesis
16.3 Orbital Floor Repair
- Recommendations for Further Research
- Conclusions
Appendices
Summary
The objective of the project was to investigate the use of embroidery techniques for the manufacture of surgical implants. The project developed a technical demonstrator in the form of a graft stent for the repair of abdominal aortic aneurysms using endovascular techniques.
The project used the demonstrator to show that fibre placement using embroidery could be applied not only to textile fibres using the CADCAM techniques of modern embroidery systems, but the placement of metallic shape memory alloy wire.
Existing fibre placement techniques were developed through the use of software and stimulated development of associated methodologies and design techniques.
In the rapidly moving world of endovascular surgery, a product requirement specification that changed throughout the project demonstrated that the technology could keep pace with the fast changing demands of the surgeons. The project has also stimulated an enhanced understanding of the problems of endovascular techniques and an MD thesis is now in preparation as a result of an underlying programme to understand attachment of the endovascular graft stent to human aortae.
- Introduction
Endovascular surgery is a comparatively new technique for the repair of abdominal aortic aneurysms.
Of the population over 65 years old, 2.4% of men have aortic aneurysms, some 0.29% of women. Using current techniques there are approximately 3,000 cases of surgical intervention each year in England and Wales. There is a 5-10% mortality rate on elective surgery, but there is a greater risk from rupture if un-operated. 10,000 per year die of ruptured Abdominal Aortic Aneurysms (AAA) in England and Wales Many patients with aortic aneurysms are too old or frail to be able to withstand open surgery. The development of satisfactory vascular stents to line the aorta that can be inserted using endoscopic techniques was essential to allow operative intervention on this group of patients.
When the project commenced in 1996, a 44 patient series of endovascular stents had been run by Professor Hopkinson at the Queen’s Medical Centre, Nottingham. There had been approximately a 25% failure rate for the current endoscopic techniques, when a change of plan during the operation required open surgery, putting the patient at significantly greater risk. Failure had generally been over an inability to maintain an effective seal between the inner surface of the artery and the outer surface of the endoscopic stent.
There had been very limited development of a number of endovascular implants in other centres in the UK , despite the potential for 12,000 – 13,000 endovascular implants per year in England and Wales subject to a satisfactory method being developed. Preliminary studies had indicated that endovascular surgery is associated with less haemodynamic derangement, which has the advantage of minimising cardiovascular morbidity and mortality.
The requirement was for an implant that would be thin enough to be introduced through a 7.3mm catheter, and having walls made from fibres that were sufficiently densely packed to provide low porosity (although this did not need to be as high as a conventional vascular prosthesis). It should also incorporate radio-opaque markers. It was required that the graft stent be arranged so as to obtain a seal against the artery wall. It was suggested that this be achieved by forming polyester strip springs at the top and bottom of the stents.
- The Demonstration component
The development of an abdominal aortic aneurysm graft stent was chosen as a demonstrator of this technology for several reasons. One of these was that endovascular surgery is a fast developing area in which one of the project collaborators had already established an international reputation. Secondly, the specification for a satisfactory graft stent provided a demanding target to be met using this new technology. Thirdly, because the novel method of manufacture (embroidery technology), overcame many of the limitations imposed by patents of other manufacturers.
- Embroidery Techniques
3.1 Cornely
There are several machine embroidery techniques in existence. Cornely embroidery uses a single needle head with a substrate material held in a pantograph, which is moved under computer control. One application of this technique in the garment industry is the tacking of heavy cord to the substrate by coiling the cord in a wrapping yarn, then stitching the wrapping yarn to the substrate with a chain stitch. This method was deemed unsatisfactory because of the lack of stitch security, (a characteristic of chain stitching).
3.2 Schiffli
This method of embroidery uses rows of needles held on a horizontal rack, with a substrate material mounted in a vertically held pantograph. The primary yarn runs partially through the thickness of the substrate and is held in place by a second interlocking yarn at the rear of the work. This approach was rejected because Schiffli machines are extremely large, high output units. A 21-yard long machine costing in the order of £1 million would produce nearly a million aneurysm graft stents per month.
3.3 Lockstitch
These embroidery machines are the most common type of commercially available equipment and are essentially adaptations of a standard lockstitch sewing machine. Lockstitching is particularly appropriate for surgical implants because the security of the stitching is very high. The machines are competitively priced, and there is a good range of software available. This commercial software for decorative embroidery is usually very much more powerful than is needed for the comparatively simple stitching needed for surgical implant applications.
Lockstitch machines are available ranging from a single embroidery head to 36 head machines. The selection of an appropriately sized machine for future mass production can therefore be made according to particular volume demands. Lockstitch embroidery machines are also highly portable. Additional capacity, should the need arise, can be provided at very short notice. As a result of this and other advantages of embroidery lockstitch machines, this route was selected for this project.
- Embroidery Software
A number of commercially available embroidery packages were examined. The package that was eventually selected is, as are all other available packages, designed for aesthetic embroidery. Similarly there are a number of levels of complexities of the software package available, typically five. The level two software was sufficiently powerful for use in this project and provided good value for money. Many of the stitch patterns used in this project required individual stitch placement rather than complex algorithms: many of the features of the software were infrequently used.
Work carried out at the University of Nottingham indicates that to satisfy the majority of patients requiring endovascular aneurysm repair, some 1,700 size variations of implant would be required. One objective of the project was to develop approaches to produce different sizes of implant using software. This was not done for two reasons: the first being that aneurysm graft sizing is the subject of an on-going study, and it has recently been learned that a considerably smaller number of grafts would need to be customised to satisfy all the patients than was previously thought. Secondly, the design of the graft stent was continually changing during the project for a number of reasons. It was decided to leave the development of very rapid design methods, using computer macros, until the design and sizing of the graft stents have reached a more stable state of development and understanding. However, a new size graft can already be developed in less than 2 hours , but it is hoped to reduce this to less than half an hour.
It has been demonstrated successfully that once a design has been completed for a stent the software design code can be transmitted by e-mail from one site to another, and manufacturing of a surgical implant may commence in less than 10 minutes.
- Endovascular Aneurysm Graft Stent Specification
The science of endovascular aneurysm repair is a rapidly evolving one. Professor Hopkinson and his team are in continual contact with other pioneering teams in this technology. Throughout the project, the requirement specification for the implant was continually updated in the light of this. However, many of the original requirements remained constant throughout the project, although the importance and the detail of some of these requirements was amended and revised. The main features of the specification are as follows:
- A base material of fine woven polyester. Industrial grade (i.e. unpigmented) polyester yarns have long been used for vascular repair and other implant materials. Microfibre polyesters, that is fibres with filaments having a count of less than 1 decitex have comparatively recently been developed, and are a preferred fibre for use in endovascular stents because of their ability to pack closely in a woven fabric and provide a high degree of leak resistance. A source of supply of a suitable fabric was identified in Italy and ultimately a large single batch of checked and certified fabric was acquired. This was sufficient to provide several years manufacturing output of the finished implant. The fabric is scoured before use to remove all spinning lubricants and other contaminants.
- A bio-inert material for the reinforcement. Conventional vascular repair textiles are reinforced using a crimping technique to concertina the fabric. However, this produces a high bulk product, which is unsuitable for insertion through a catheter. Therefore the fabric must be straight walled with a reinforcement material. This prevents the lumen closing if the graft is required to pass through a tortuous diseased artery.
- High columnar strength. Concern was expressed that the graft might collapse under pulsatile blood pressure and move within the artery. Such columnar strength would also aid insertion and minimises any possibility of lengthways collapse of the graft. Similarly, it was expected that the strength of the column would reduce the possibility of the occlusion of the lumen in a twisted artery.
- A simple but effective anchoring system at the top of the graft against the healthy artery above the diseased section to prevent the device migrating. The method of achieving this fit should be either a hook or similar system or a tight spring fit against the artery wall.
- A bulk sufficiently low that the whole device would pass through a 22 French catheter (7 mm diameter).
- A device that would emerge from within the catheter and deploy rapidly as required within the aneurysm, and provide a seal against the vessel wall to prevent blood leakage around the device (an endoleak).
- Implant Materials
It was originally envisaged that all the embroidery work would use conventional suture materials for use as the implant embroidery thread. These products, manufactured by one of the collaborators, were considered to be ideal because of the ready acceptance by surgeons of such materials. Materials used during the project included conventional braided polyester suture thread together with nylon and polyethylene monofilament. Additionally, non-implant grade polypropylene was used to try to attain the required stiffness. These aforementioned materials were not fully successful, so superelastic alloy was used. Nitinol alloy has been used with considerable success in other implant devices. Although there is ongoing discussion about its biocompatibility, the extremely rapid oxidation of the metal provides an oxide barrier between the metal and the bio-system. Any long-term risks associated with nickel ion release are thought to be minimal in this patient group.
- Reinforcement Patterns
There were a very considerable number of ideas for reinforcement. Some of these were much more successful than others; the final design for the implant was arrived at by an iterative process that resulted in 153 designs having been produced. Many of the design ideas are shown in the patent for the endovascular stent.
The design eventually adopted was a «ladder pattern» which not only provided sufficient reinforcement of the polyester textile to prevent the lumen collapsing, but also provided sufficient columnar strength to achieve what was required in that direction. The Nitinol wire was preshaped and set, and then precisely stitched in place on the base cloth using conventional lockstitch embroidery methods. It was found later in the project that the spring fit of the device against the wall of the healthy aneurysm above and below the damaged section of artery could be better achieved if the spring section was placed inside the completed stent. By stopping the embroidery programme at an appropriate stage, the Nitinol wire is readily fed through the base cloth to emerge externally before continuing the stitching process.
After the embroidery takes place, including attaching wire and hooks to the base cloth, the tube of the finished stent is completed by hand stitching. Because of the low diameter of the finished stent, it is very difficult to use secure stitching methods to form a narrow tube. Some research has been carried out to attempt to identify a method of machine attachment. It is noteworthy that no secure stitching method that can be applied to this problem has been patented in the last 160 years.
- Processing
After the graft stent is closed into a tube, the device is scoured and sterilised using gamma irradiation. It is then ready for loading into the implantation device.
- Project collaborators
The consortium comprises a manufacturer experienced in the high volume production of textile surgical applications, a small research orientated manufacturer experienced in the interface between textiles and clinical surgery; and research orientated clinicians with experience of the applications for the demonstration product planned.
9.1 Ellis Developments
Ellis Developments Limited was founded in 1985, and specialises in advanced textile technology, particularly the development of textiles for engineering and surgical applications. The company was responsible for the textile design of Europe’s best selling artificial ligament, the Leeds-Keio Ligament for the replacement of anterior cruciate ligament of the knee. They previously had administered a £700,000 LINK Structural Composites project, to apply embroidery to composite preform production and have won SMART awards in 1988 and 1999. Ellis Developments Limited is an SME.
9.2 Pearsalls Sutures
Pearsalls Sutures were a division of Bridport Gundry plc and were founded in 1795. They were subsequently owned by the US based Marmon Corporation. Pearsalls have been making suture threads for over 100 years, and supply most independent suture companies, including Davis and Geck, B. Braun and US Surgical. Over 90% of their work is exported and they have won a Queen’s Award for Export.
9.3 Division of Vascular Surgery, University of Nottingham
The Division of Vascular Surgery under the leadership of Professor Brian Hopkinson at the University of Nottingham is part of the Department of Medical and Surgical Sciences. Professor Hopkinson has an international reputation for his work in endovascular surgery and has carried out the largest number of endovascular procedures in the world.
9.4 Anson Medical Limited
Anson became actively involved in the project in May 1997, when they were brought in to advise on the use of shape-memory alloy. Although they did not become an official partner, Anson attended project meetings as well as becoming heavily involved in the project. An agreement was signed in July 1998 whereby Anson committed development resources to commercialise the AAA graft stent. They have been approached by a number of leading companies who wish to distribute the product in specific territories.
Anson Medical provided technical input in the form of providing pre-shaped ladders of shape memory alloy and a large variety of technical advice and assistance. They also provided significant funding for parts of the project for which the budget was insufficient. Anson Medical Limited is an SME.
9.5 Contributions of each Partner
Ellis Developments Limited have considerable expertise of textile surgical implants and embroidery. They acted as interface between textile and clinical disciplines, and to lead intellectual property aspects, as well as being project managers.
Pearsalls Sutures provided controlled facilities for the operation of embroidery machinery and know-how on the handling, production and application of textile materials. They installed a class H clean room during the project and are now registered to ISO 9001 and EN 46001.
The Division of Vascular Surgery at the University of Nottingham provided expertise in endovascular stenting, having run the world’s largest series of endovascular surgical procedures. They had previously developed an understanding for the requirements of effective stent that can be applied to patients who are too old or too frail to withstand abdominal surgery to treat aortic aneurysms.
9.6 Interaction of collaborators
There were formal project meetings held each quarter for the duration of the project, which reviewed work carried out during the previous period, and planned future work. Each 6 months a full project meeting was attended by representatives of the programme managers, Quotec and project monitors from the Department of Health. These minuted meetings were supported by regular meetings, telephone calls, faxes, e-mails, and informal meetings between partners.
- Design Control
By September 1999 good evidence had been provided that the Nitinol-polyester graft combination goes into a normal sheep aorta satisfactorily. It fixes well with hooks, and seals satisfactorily as far as experimental aneurysms are concerned. The limited amount of histology carried out then shows no adverse reaction at all. At that time there were 7 sheep with stent-graft and aneurysms in place and an additional 2 sheep had stent grafts in their aorta, one without an aneurysm and one with an aneurysm had been sealed off, despite having made an attempt to produce a forced endoleak.
The next stage was to prove that the graft would follow tortuous vessel paths without kinking which was one of the first requirements for the graft. A 20 cm long graft was made and put in as an open procedure through an ateriotomy with a 3600 curl in it. Full patency of this graft was maintained in two sheep for over 24 hours.
- Endoleak Research
From one of the animals sacrificed, extremely good evidence was provided that a small endoleak can enable systolic pressure to enter the aneurysm. The presence of clot within the aneurysm prevents the blood getting back into the aorta and maintains a mean intra-aneurysm pressure much higher that the systemic mean arterial pressure. This very interesting observation provides an explanation as to why a small endoleak is very often fatal to patients, sometimes within a week of surgery. Endoleaks have been a major problem in endovascular surgery and as they are the greatest source of morbidity and mortality a much better way of sealing was thought essential.
It may be that combinations of thin nylon or polyester sacks inserted within the graft and then inflated with silicone rubber could help provide a very good seal. Alternatively sacks of thin compliant nylon or polyester could be fed into the endoleak from the lumen of the aorta and then inflated to seal off the affected area. A request was therefore put in to enable the study of another 3 sheep with the 3600 curled graft in them and 3 more with endoleaks that could be sealed by an intra-aneurysmal bag of silicone rubber. This work, outside the project, continues.
Before proceeding to the endoleak experiments in animals, a bench model was developed to try to demonstrate what size of endoleak is required to raise pressure in the aneurysm above the mean arterial pressure. It is proposed that implantable pressure sensors will then be put inside the experimental aneurysms. External monitoring will be used over a period of several months to check the pressure and pressure wave within the aneurysm in live sheep.
- Cost Evaluation
There is an ongoing debate about the comparative cost benefits of endovascular aneurysm repair techniques as against conventional open repair. The cost of current endovascular repair devices is very high, but it is expected that partly as a result of the comparative speed and simplicity with which customised devices can be produced using embroidery techniques, the Nottingham Graft Stent will be available to the NHS at a marked discount to current prices.
- Achieved Project Deliverables
- An understanding of the design rules for embroidered surgical graft stents.
- An understanding of design rules for other textile surgical implants.
- The development of validated manufacturing methods for the design and manufacture of custom made implants.
- A demonstration of extremely rapid prototyping and manufacturing speeds.
- Structural design methods for embroidered textile reinforcement
- Demonstration for potential for embroidery as a method of manufacture of customisable surgical implants
- Manufacturing method for surgical implants which is highly flexible, has improved structural performance and has high consistency compared with conventional textile implant manufacturing techniques.
- Established design control procedures
- Established manufacturing control procedures.
- Apparently successful animal trials of the demonstration product.
- Apparently successful laboratory testing of the demonstrated product.
- Developed protocol and software design rules.
- Validated implant design.
- Product almost ready for submission to Medical Devices Agency.
14 Dissemination
14.1 Published Conference Papers
Ellis J.G, Wallace W.A, Neumann L, Textile For the Repair of The Rotator Cuff of the Shoulder, Medical Textiles’ 96, Bolton UK
Hopkinson BR, Macierewicz J, What should we look for in the next generation of endovascular grafts? 26th Annual Symposium on Current Critical Problems, new horizons and techniques in vascular and endovascular surgery, New York November 1999
Hopkinson BR, Macierewicz J, Beaton G. Combined interventional and surgical treatment of abdominal aortic aneurysms using nickel-titanium tubular grafts – an alternative approach. In: Medical Applications for Shape Memory alloys. Seminar: 2nd September 1999. Pub: IMechE ISBN 1860582419
14.2 Conference Presentations
Ellis J.G Surgical Implants Using Embroidery Techniques UK liaison committee for Sciences Allied to Medicine and Biology, Medical and Biology New Technology, 4th International Conference, Brunel, 1999
Ellis JG Engineering and Surgical Textiles by Embroidery, World Conference 2000, The Textile Institute, Manchester, April 2000.
Ellis J.G & Hopkinson B.R, The Endoscopic Repair of Abdominal Aortic Aneurysms Using Embroidered Textiles. Link Medical Implant Seminar, Meriden, UK, 1988
14.4 Thesis
J Macierewicz. Study on the development of an embroidered prosthesis for endovascular repair of abdominal aortic aneurysms. MD Thesis
14.5 Others
Display at Tomorrow’s World Live Exhibition, NEC Birmingham, July 1999
14.6 Patents
PCT International Patent Application WO99/37242 Reinforced Graft
European Patent application – Device for the Reinforcement of the Rotator Cuff of the Shoulder
US Patent 5990378 – Textile Surgical Implants
European Patent Application 96303741.1 – Textile Surgical Implants
European Patent 074416282 «Textile surgical Implants»
US Patent 98/652316 «Textile Surgical Implants»
«Vascular Graft Stent»
- Exploitation
15.1 Achievements and Further Plans
Anson Medical Ltd have taken rights to distribute the AAA Graft Stent across the world. They have been approached by a number of leading companies who wish to distribute the product in specific territories.
Once the product has been submitted to and approved by the Medical Device Agency, the product will be available for clinical tests. Initially these will be carried out at Queen’s Medical Centre, Nottingham.
A number of other embroidered implants are in various stages of development in parallel with this project. These include several shoulder repair devices, which have already been used on a small number of patients, and a prosthesis for the replacement of an intervertebral disc of the cervical spine.
- Parallel Projects
Concurrent with the project, there have been a number of developments taking place using embroidery techniques. These include the following:
16.1 A device for the Repair of the cervical spine.
This is the embroidery of a complex textile shape, which fits around a silicone cervical spinal disc prosthesis. The textile is designed to replace the spinal ligaments and hold the disc prosthesis firmly in place. Ten million cycle testing has demonstrated that the device retains its patency after such a repeated load and unload cycle. The device was developed in conjunction with Mr Andre Jackowski of the Royal Orthopaedic Hospital, Birmingham. A patent has been filed and published for this device.
16.2 Shoulder Prosthesis
Three shoulder products developed in conjunction with Professor W A Wallace and Mr Lars Neumann of the Shoulder and Elbow Unit, at Nottingham City Hospital/University of Nottingham. There have already been a number of implantations made under special exemption rules, although CE marking have not yet being possible on the product. Two other shoulder devices are in an advanced stage of development, one of which has been patented.
16.3 Orbital Floor Repair
Designs have been developed for the repair of the orbital floor for use by Maxillofacial surgeons. Some of this work has been done in conjunction with the AMPICS project funded by the EPSRC and based at the University of Nottingham. The device, made from polyester yarn, meets all the criteria of the surgeon, Mrs Sheila Fisher of the Oral Maxillofacial Unit, Nottingham, with the exception that she requires a biodegradable raw material. Existing biodegradable raw materials such as polylactic acid and polygycolic acid degrade far too quickly for use in this particular application. Following a successful trial using Ethicon’s slowly biodegradable Panacryl fibre in the shoulder, it is hoped that Ethicon will release sufficient fibre for tests to be carried out using this product
- Recommendations for Further Research
Development of an embroidered graft without seam
Development of an endoleak model bench-test
Development of system of external monitoring of pressures inside the aneurysmal sac
Study on filling the aneurysmal sac with various thrombogenic substances as a way to prevent endoleak as well as provide external support for the stent graft.
Study on deformation of graft during and after deployment.
Development of other surgical implants using embroidery techniques.
Development of a seam closure sewing machine for small diameter tubes.
Development of software for the rapid design of AAA graft Stents.
- Conclusions
This project which took rather longer than originally planned, mainly because of circumstances beyond the control of the collaborators, has developed a demonstration device which shows every prospect of being technically highly successful. The technical success is likely to be reflected in the commercial success of the device, the signs for which are extremely encouraging at the time of writing. There are still many unsolved surgical problems for the endovascular repair of abdominal aortic aneurysms, but plans exist to meet these by continued development programmes. Many of the further developments are expected to be funded through income generated by sale of the first stage graft design.
- Acknowledgements
We acknowledge with thanks the help of all members of the team that made this project so successful. These include: –
University of Nottingham
Dr Jean-Noel Albertini
Mr Bruce Braithwaite
Mr David Edwards
Dr Nabil El-Marasy
Dr Stravros Kaliapor
Dr Jan Macierewicz
Mr Mark Parry
Mr Ron Walker
Mr Stuart Walker
Mr Waquar Yusuf
Pearsalls Limited
Mrs Elizabeth Buller
Dr Alan McLeod
Mr Lawson Lyon
Ellis Developments Limited
Miss Pauline Ellis
Anson Medical Limited
Dr Peter Philips
Dr Gail Beaton
Department of Health
Mr Anthony N Horn
Mr Jonathan Bickley
Quo-Tec Limited
Mr Matthew Chapman
Appendix 1
Collaborators
Pearsalls Sutures
Dr Alan McLeod
Mr D Lawson Lyon
Ellis Developments Limited (SME)
Julian G Ellis
Mr Peter W Butcher
University of Nottingham, Department of Vascular Surgery
Professor Brian R Hopkinson
(Anson Medical Limited, Didcot, Oxfordshire joined the project informally, providing expertise in shape memory alloys and as the preferred company to market the product)
Appendix 2
Project Objectives
To produce a validated method for the design and cost effective production of low and medium volume production textile surgical implants.
To develop a specific application in the area of vascular surgery in order to demonstrate the technique
A set of design rules for embroidery of implants
Reproducible method of manufacture to meet the requirements of good manufacturing practice
Development of manufacturing conditions and process appropriate to implant production
Method validated through clinical tes
All these targets have been achieved, although time has not permitted the clinical testing of the device produced. This was mainly due to a considerable delay in obtaining Home Office approval for the animal work. However, the device is almost ready for submission to the Medical Devices Agency for approval for clinical test to commence.
Back to top
Implant Picture Gallery
Back to top

Back to top
Address
1, Fair A Far, Cramond
Edinburgh,
Scotland
EH46QB
Contact Us
(+44) 07759248863
michelle@raedevelopments.com